Meaningful engagement is central to patient-centered health research but implementing it at a national scale can pose challenges—from recruiting participants and sustaining involvement, to ensuring patient perspectives shape research questions and outcomes. Research teams can use resources available through PCORnet®, a national patient-centered health research network, to enable large-scale research. A recent supplement in the journal Medical Care highlights how two PCORnet® Study teams leveraged the PCORnet infrastructure to improve recruitment and retention efforts, and to embed engagement throughout the research lifecycle.
The CHI-RON Study: Improving Representation Through Data-Driven Recruitment
Working with 12 sites across four PCORnet® Clinical Research Networks, the CHI-RON Study examined the effects of gaps in recommended care for individuals with adult congenital heart disease (ACHD).
CHI-RON, or Congenital Heart Initiative–Redefining Outcomes and Navigation to adult-centered care, is one of more than 60 PCORnet® Studies answering critical patient-centered questions on heart disease, metabolic conditions, obesity and more.
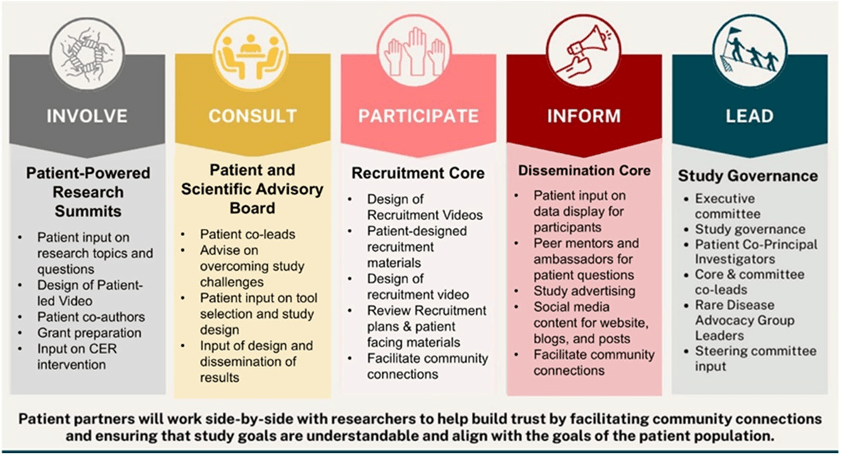
Repurposed from article, available here online.
CHI-RON included two patients as co-investigators, a role that allows embedded patient input at every stage. Additional patient partners also provided input on the study design, advised on challenges, designed recruitment videos, facilitated community connections, and created social media content.
When asked how they felt about their contributions to the research, the patient partners reported strong feelings of meaningfulness and contribution (measured using the validated Patient Engagement in Research Scale; PEIRS22).
“Building a truly patient-centered research team in which patient partners were included at every step enabled us to create a study that drew people in,” said Ruth Phillippi, MS, patient co-investigator and program lead at the Washington Adult Congenital Heart Program. “Not just to the registry but to a distinct and engaged research coalition that values our opinions and lived experiences.”
The CHI-RON study team also used the PCORnet® Common Data Model to help increase diversity in the trial.
Recruiting participants that reflect real-world populations has been a persistent challenge in ACHD research. Individuals who are younger, male or not actively engaged in clinical care have historically been underrepresented, limiting generalizability.
To address this, the CHI-RON study team developed a systematic recruitment approach. Patient, clinician, and researcher partners worked with the data science teams within PCORnet to design an algorithm to identify individuals traditionally not reflected in ACHD research and generated site-specific recruitment lists to support tailored outreach efforts. This tailored approach, embedded in the study design, successfully expanded demographic reach.
“I really do think it was the combination of the recruitment algorithm in addition to our engagement strategy that helped successfully increase the number of patients that we had previously underrepresented,” Anitha John, MD, PhD, CHI-RON investigator, reflected during a recent PCORnet® Best Practice Sharing Session.
“We were able to increase the numbers of younger, male patients, and also achieved better representation across race, ethnicity, and educational background.”
Read the full publication here.
PRECIDENTD: Sustaining Engagement in a National Pragmatic Trial
The PCORnet® Study PRECIDENTD (PRevention of CardIovascular and DiabEtic KidNey Disease in Type 2 Diabetes) is a national trial comparing two classes of diabetes medications, SGLT-2and GLP-1 (sodium-glucose cotransporter-2 inhibitors and glucagon-like peptide-1 receptor agonists).
Large pragmatic trials often struggle with retention and adherence, particularly when resources are limited and study teams are distributed across many sites.
“Drugs in these classes can be expensive and insurance coverage varies,” said PRECIDENTD investigator Lindsay Mayberry, MS, PhD. “That means that patients often needed additional support in order to actually adhere to trial requirements and complete study assessments. Patient input helped us understand the challenges patients face and how we might get ahead of them.”
PRECIDENTD engaged patients to co-design an automated text message-based tool designed to identify participants who need support to adhere to study medications, as well as to remind participants of study timelines and follow-ups, and express appreciation for their role in research. This included holding a community engagement studio to receive input on the study design and to shape the text messaging program’s goals and structure. Researchers then collaborated with a patient advisory council to codesign message content and tone and ensure the texting intervention reflected the real-world needs and experiences of people living with type 2 diabetes. In addition to helping to test the system, two patient partners served in decision-making roles on the executive committee alongside study investigators and helped to publish and disseminate results.
“Pragmatic trials place real demands on participants and study staff alike,” said patient partner Ed Simeone. “As a patient partner, being part of the co-design process from the start — including the development of the text messaging program — helped ensure that communication was clear, burden was manageable, and participants felt genuinely supported. That kind of collaborative approach builds trust and turns a research study into a shared endeavor.”
The program achieved an 80% response rate to text messages, with 25% of these interactions identifying barriers to taking the study medication that required coordinator support. By targeting outreach to those most in need, the approach helped study teams use limited resources more effectively while keeping participants engaged.
Through targeted outreach and other engagement strategies, PRECIDENTD reflects the comprehensive approach to engagement that all PCORnet® Studies should follow.
Read the full publication here.
These two PCORnet® Studies demonstrate some of the ways researchers can leverage the research-ready community and expertise within PCORnet to deepen engagement efforts.
Explore the Medical Care Supplement
The publications highlighted here are part of the recent Medical Care supplement, “PCORnet®: Accelerating Patient-Centered Comparative Clinical Effectiveness Research.” The supplement includes 19 peer-reviewed, open-access articles showcasing how investigators are using PCORnet to reshape patient-centered health research. Learn more and explore the full supplement here.
Contact the PCORnet® Front Door to learn how the PCORnet infrastructure can support your patient-centered health research.
