PCORnet® Playbook
Introduction to PCORnet®
PCORnet is one of the largest real-world data networks in the country for patient-centered research, with a proven history of supporting hundreds of high-quality research studies. This network is a valuable resource designed to make health research more efficient, especially in patient-centered comparative clinical effectiveness research (CER). PCORnet connects a wide range of health data, research expertise, and patient perspectives to improve the nation’s ability to answer critical health questions. It was developed with funding from the Patient-Centered Outcomes Research Institute® (PCORI®).
More than a decade ago, researchers across the United States posed an important question:
“What if patients and caregivers, researchers, health systems, and clinicians across the U.S. could work together on research that matters to patients?”
For too long, traditional clinical research has struggled with inefficiency and separation. Studies often take too much time, involve overly complicated steps, and don’t focus on the questions that matter most to patients. Many research projects have been shaped by academic or business priorities rather than the real-world concerns of patients and their caregivers. This disconnect means that the results are often less useful in addressing everyday health issues.
In 2013, PCORnet was developed with funding from PCORI to change how research is done. The PCORnet infrastructure brings together patients, researchers, and healthcare systems to collaborate, share data, and make decisions together. By focusing on teamwork, it ensures that everyone’s voice—especially the voice of patients—is heard in the research process.
PCORnet links information from the electronic health records (EHRs) of over 47 million people across the country, along with insurance claims and surveys where patients share their experiences. This means researchers can include groups of people in their studies and design projects that represent different communities. For example, PCORnet made studies like ADAPTABLE (a heart health study comparing aspirin dosages) and RECOVER (researching long COVID) possible. These studies were not only large-scale but also focused on questions that impact people’s lives.
With a collaborative infrastructure and powerful tools, PCORnet allows researchers to break down the barriers that have held research back for so long. That means researchers can work faster, include more people, and deliver results that truly help patients and caregivers in their daily lives.
Last Updated: May 18, 2026
Contents
- What Makes PCORnet Unique?
- The PCORnet® Common Data Model
- The PCORnet® Common Engagement Model
- Understanding the Infrastructure of PCORnet
- PCORnet® Clinical Research Networks
- The Coordinating Center for PCORnet®
- Patient Partners
- Governance Teams for PCORnet
What Makes PCORnet Unique?
The PCORnet infrastructure stands out from other distributed networks because it combines high-quality data, research expertise, and patient partnership to support impactful patient-centered health research. For over a decade, this proven infrastructure has successfully supported studies funded by federal, non-profit, and industry partners, exhibiting capability and reliability as a national resource. PCORnet demonstrates that developing shared infrastructure can deliver:
- National-Scale Collaboration: Facilitates studies that enable national-scale, multi-institutional collaborations.
- Patient-Partnered Research on Questions that Matter: Ensures research questions are meaningful to patients and caregivers and that outcomes are scientifically rigorous.
- Public Accessibility and Flexibility to Answer a Range of Research Questions: Provides services to guide interested researchers and partners, regardless of experience level, including non-traditional researchers such as patient and community partners, frontline clinicians, and caregivers. PCORnet supports studies of all sizes, from small to large multi-center projects, as well as a variety of study designs, including observational and interventional research.
10+
Years of Patient-Centered Research
Data as of February 2026

The PCORnet® Common Data Model
Data from individual PCORnet® Clinical Research Networks (CRNs) are standardized using the PCORnet® Common Data Model (CDM). The PCORnet® CDM is a foundational element of the PCORnet infrastructure that brings EHRs, claims, and other data sources from all eight PCORnet® CRNs into a common language that can be integrated and analyzed. Rigorous quality checks through data curation are a cornerstone of the CDM, ensuring that data are reliable, accurate, and suitable for meaningful research. By applying consistent data definitions and structures, the PCORnet® CDM facilitates national-scale, multi-center research that allows you to draw meaningful insights across populations.
In the United States, few research networks operate on the scale of PCORnet while also using a CDM to standardize data across sites. While this unlocks exciting opportunities for national-scale research, challenges remain when working with information not originally collected for research, such as variations in clinical documentation, missing data, errors, and biases. These limitations are inherent to all research using data collected during healthcare delivery. However, PCORnet® CRNs are well-equipped to help you navigate these complexities and address data considerations effectively.
Learn more about using real-world data for research in the Living Textbook of Pragmatic Clinical Trials
Watch these videos for more information on the challenges of using healthcare data and the systems designed to address them so researchers can answer important questions.
The Big Picture: Healthcare Data and Interoperability
Common Data Models
The PCORnet® Common Engagement Model
The PCORnet® Common Engagement Model establishes network-wide engagement standards, making it easier for investigators—especially those new to PCORnet and partner-engaged research—to navigate the research process. By setting clear, foundational engagement expectations, this model helps reduce barriers to participation, ensuring that research powered by the PCORnet infrastructure is accessible and impactful.
Understanding the Infrastructure of PCORnet
The PCORnet infrastructure is a collaborative ecosystem where PCORnet® CRNs, Patient Partners, and the Coordinating Center for PCORnet® work together to enable efficient, national-scale patient-centered health research. Each part of the infrastructure plays an important role in fostering collaboration, streamlining processes, and ensuring that high-quality data and patient partnerships are at the core of every study.
PCORnet® Clinical Research Networks (CRNs)
The PCORnet infrastructure includes eight large PCORnet® CRNs across the United States. PCORnet® CRNs leverage insights from everyday health encounters with over 47 million people each year. EHR data from more than 13,000 sites of care, including ambulatory and inpatient care, academic medical centers, hospitals, community clinics, and other healthcare providers is the foundation of PCORnet. For some studies, administrative claims or other records representing care received outside the healthcare delivery system are available. The expansive breadth of these networks contributes to meaningful research.

>47 Million in the Network
PCORnet represents data from everyday healthcare encounters with more than 47 million people across the U.S. each year.

*The data density map illustrates single addresses that are associated with individual PCORnet® CRN encounters.
Is PCORnet Representative?
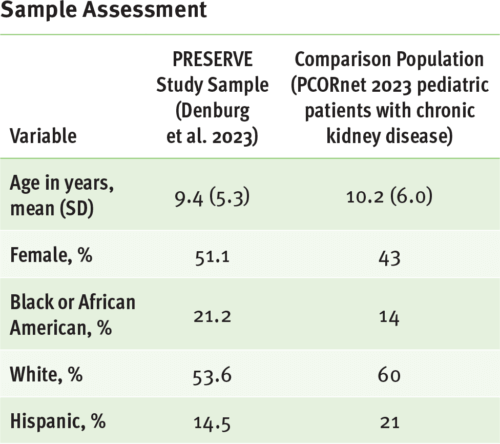
 A key strength of PCORnet is that the patient population accessible through the network is broadly representative of the U.S. population across age, sex, race, and ethnicity. This makes it a valuable resource for studies focused on people who receive healthcare services in the U.S. However, it's important to recognize that PCORnet data resources primarily represent people who have access to healthcare services, which can be influenced by factors such as socioeconomic status and geographic location. Furthermore, the representativeness of any study using PCORnet will depend on its specific eligibility criteria, clinical focus, population of interest, and design and enrollment features.
A key strength of PCORnet is that the patient population accessible through the network is broadly representative of the U.S. population across age, sex, race, and ethnicity. This makes it a valuable resource for studies focused on people who receive healthcare services in the U.S. However, it's important to recognize that PCORnet data resources primarily represent people who have access to healthcare services, which can be influenced by factors such as socioeconomic status and geographic location. Furthermore, the representativeness of any study using PCORnet will depend on its specific eligibility criteria, clinical focus, population of interest, and design and enrollment features.
Over the last decade, PCORnet® CRNs have built strong partnerships and developed a deep understanding of their data, allowing sites participating in PCORnet to confidently assess whether their data is fit for a particular research purpose. This longstanding collaboration enables PCORnet® CRNs to support the conduct of national-scale, multi-center studies that prioritize patient-centered outcomes with efficiency and accuracy. You may also be able to use PCORnet resources with data from other research sites that are not partnered with PCORnet, enabling a more comprehensive analysis. This combined approach allows for larger study populations and enhanced ability to answer complex clinical questions.
PCORnet® CRNs

The Coordinating Center for PCORnet®
PCORnet® CRNs are united by the Coordinating Center for PCORnet®, which serves as the organizational and communication hub of PCORnet. It offers services to investigators and research teams, patient and community partners, and the public. The Playbook topic, Power Your Research provides details about PCORnet services including Study Feasibility Reviews, Network Collaborator Requests, and Data Network Requests. These services maximize coordination and the accessibility of the PCORnet infrastructure. The Coordinating Center is home to the PCORnet® Front Door, which serves as the access point for the PCORnet infrastructure. It also facilitates meaningful engagement with patients, clinicians, and researchers and leads communication efforts to share updates, promote findings, and raise awareness of PCORnet.
Patient Partners
At the heart of all patient-centered health research efforts is meaningful engagement of people who have lived experience. Each PCORnet® CRN includes Patient Partners, and every PCORnet® Study requires that patients’ needs and perspectives are included in the research process. Patient and caregiver representatives are also included in overall governance as members of the PCORnet® Steering Committee. Other communities engaged as part of research include frontline clinicians and staff, health system leaders, payers, and policymakers.
Benefits of this deep engagement infrastructure include:
- Insights into the real-life impact of a health condition, helping you better understand its effects and the challenges patients face as you prepare your research.
- Study designs that minimize burden to participants, making it easier for people to take part and boosting enrollment.
- Research goals that align with what truly matters to patients, leading to results that can enhance their quality of life.
- Collaboration between researchers and patient communities, encouraging participation in clinical trials.
- Communication of research findings in clear, accessible ways that patients and their caregivers can understand and use to guide decisions about their healthcare.
Even for those who recognize the benefits of patient engagement, putting it into action can be challenging. The PCORnet infrastructure reduces these barriers by offering engagement support and expertise. The PCORnet® Front Door team can advise on engagement strategies, provide resources like recruitment toolkits or patient handbooks, and help you connect with its extensive network of experienced partners who are enthusiastic about contributing to research.
In its first decade, the PCORnet infrastructure has made great strides in helping researchers embed the patient voice at every level. We are not experts at clinical research, but we are experts in what it is like to be a patient considering participation in a clinical trial—and for a research team designing a study, that expertise can be quite valuable.
— Greg Merritt, Patient Partner on the PCORnet® Steering Committee
Governance Teams for PCORnet
The PCORnet® Steering Committee and Executive Management Team guide PCORnet operations to ensure patient-centeredness and effective decision-making for this national resource. Patient representation within these groups ensures that research is meaningful and aligned with patient needs.
The Steering Committee promotes efficient, compliant, and open science practices, supports accessible research tools, and prioritizes high-quality data, while the Executive Management Team steers strategic priorities. Additional committees and workgroups contribute specialized expertise to enhance governance and decision-making.
What Kind of Research Can the PCORnet® Infrastructure Support?
PCORnet is a versatile and valuable national resource that can support a wide range of research activities.

Real-World Evidence Research
 Real-world evidence studies use data collected from routine health visits and other real-world settings to generate insights that can inform healthcare decisions. Using the vast amount of data available through PCORnet, you can conduct observational studies, assess the effectiveness and safety of treatments, and learn about the natural history of diseases. These types of studies are crucial for understanding how interventions work in different patient populations and real-world healthcare settings, providing valuable information that complements traditional clinical trials.
Real-world evidence studies use data collected from routine health visits and other real-world settings to generate insights that can inform healthcare decisions. Using the vast amount of data available through PCORnet, you can conduct observational studies, assess the effectiveness and safety of treatments, and learn about the natural history of diseases. These types of studies are crucial for understanding how interventions work in different patient populations and real-world healthcare settings, providing valuable information that complements traditional clinical trials.
Comparative Clinical Effectiveness Research
 Comparative clinical effectiveness research (CER) compares two or more interventions or strategies to prevent, diagnose, monitor, or treat a health condition. By comparing options that have been proven to work or are commonly used, CER helps patients, clinicians, and policymakers make informed decisions about health and healthcare. The extensive data resources and collaborative network available through PCORnet enable you to conduct multi-center CER studies at a national scale, providing strong evidence on the relative effectiveness of different interventions. Through access to large populations, you are also able to design studies based on real-world treatment patterns, better recruit patients with specific health conditions, understand various treatment effects, and combine health outcomes data with patient-reported outcomes.
Comparative clinical effectiveness research (CER) compares two or more interventions or strategies to prevent, diagnose, monitor, or treat a health condition. By comparing options that have been proven to work or are commonly used, CER helps patients, clinicians, and policymakers make informed decisions about health and healthcare. The extensive data resources and collaborative network available through PCORnet enable you to conduct multi-center CER studies at a national scale, providing strong evidence on the relative effectiveness of different interventions. Through access to large populations, you are also able to design studies based on real-world treatment patterns, better recruit patients with specific health conditions, understand various treatment effects, and combine health outcomes data with patient-reported outcomes.
Population Health Research
 Population health research focuses on understanding the health outcomes of groups of people and the factors that affect those outcomes. The data sources available through PCORnet are ideal for conducting this type of research. By analyzing data on social factors, healthcare utilization, and health outcomes, you can identify patterns to inform population health interventions and policies.
Population health research focuses on understanding the health outcomes of groups of people and the factors that affect those outcomes. The data sources available through PCORnet are ideal for conducting this type of research. By analyzing data on social factors, healthcare utilization, and health outcomes, you can identify patterns to inform population health interventions and policies.
Pragmatic Research
 Pragmatic research evaluates how well a potential treatment or strategy works in real-world healthcare settings, as opposed to the controlled environments of traditional clinical trials. These studies provide evidence that can be directly applied to everyday clinical practice. The PCORnet infrastructure supports pragmatic research by facilitating the collection and analysis of real-world data, enabling you to assess the impact of interventions in routine care settings. The infrastructure also enables research designs that are embedded in care settings.
Pragmatic research evaluates how well a potential treatment or strategy works in real-world healthcare settings, as opposed to the controlled environments of traditional clinical trials. These studies provide evidence that can be directly applied to everyday clinical practice. The PCORnet infrastructure supports pragmatic research by facilitating the collection and analysis of real-world data, enabling you to assess the impact of interventions in routine care settings. The infrastructure also enables research designs that are embedded in care settings.
Health Systems Research
 Health systems research examines how healthcare delivery systems work and how they can be improved to enhance healthcare quality and efficiency. By studying various aspects of healthcare systems, such as care coordination, patient engagement, and health information technology, the PCORnet infrastructure makes it possible to identify best practices and develop strategies to improve healthcare delivery.
Health systems research examines how healthcare delivery systems work and how they can be improved to enhance healthcare quality and efficiency. By studying various aspects of healthcare systems, such as care coordination, patient engagement, and health information technology, the PCORnet infrastructure makes it possible to identify best practices and develop strategies to improve healthcare delivery.
Implementation Science Research
 Implementation science studies examine how best to help healthcare delivery settings implement an effective intervention or evidence-based practice, thereby improving the quality of care. The goal of these studies is to identify factors that influence successful uptake of research findings within a broader community. Results of implementation science studies inform rollout efforts of improved practices on a wide scale.
Implementation science studies examine how best to help healthcare delivery settings implement an effective intervention or evidence-based practice, thereby improving the quality of care. The goal of these studies is to identify factors that influence successful uptake of research findings within a broader community. Results of implementation science studies inform rollout efforts of improved practices on a wide scale.
As a massive research network that can capture information from many healthcare systems, PCORnet had an important role during the pandemic response. Via PCORnet, the CDC and others learned detailed surveillance information that was essential to help our leaders, institutions, and patients understand and combat the pandemic.
— Tom Carton, Principal Investigator at REACHnet, a PCORnet® Clinical Research Network
Frequently Asked Questions
No, PCORnet® Studies are supported by a range of funding sources. Besides PCORI, other funders have included the National Institutes of Health (NIH) and companies in the pharmaceutical and biotech industries. Use of PCORnet (e.g., for a feasibility query) does not guarantee future PCORI funding for the research.


 Sources:
Sources: