PCORnet® Playbook
Using the Power of PCORnet® for Patient-Centered Health Research
This section includes examples of how research teams have used the PCORnet® infrastructure to accelerate evidence generation and translate healthcare innovations into population health benefits.
PCORnet® Studies span a range of sizes, designs, therapeutic areas, populations, and funding sources, including the Patient-Centered Outcomes Research Institute® (PCORI®), the National Institutes of Health (NIH), and industry sponsors. More than 300 research projects have used the PCORnet infrastructure to conduct patient-centered health research that helps patients, caregivers, clinicians, health systems, and policymakers make better-informed decisions to improve health and healthcare.

Overview of the PCORnet® Infrastructure
The PCORnet infrastructure enables insights from high-quality health data, patient partnership, and research expertise to deliver fast, trustworthy answers that advance health outcomes. The case studies in this section illustrate how PCORnet® Study teams successfully leverage these assets – individually and in combination – to improve their research.
 The robust infrastructure of PCORnet, and particularly the PCORnet® Common Data Model that unites millions of disparate data points so they are usable and meaningful for research, gave us a significant head start in our search for answers.
The robust infrastructure of PCORnet, and particularly the PCORnet® Common Data Model that unites millions of disparate data points so they are usable and meaningful for research, gave us a significant head start in our search for answers.
— Mark Pletcher, Principal Investigator,
BP Control Laboratory, a PCORnet® Study
Overview of Case Studies
The table below provides general information about each study included in this section. This is a small sample of the wide range of research activities supported by the PCORnet infrastructure. These examples include some of the largest clinical research studies conducted in the U.S. for specific populations or conditions.
| Study Name | Design | Population | Therapeutic Area | Funder |
|---|---|---|---|---|
| Aspirin Dosing: A Patient-Centric Trial Assessing Benefits and Long-Term Effectiveness (ADAPTABLE) |
Intervention Trial | 15,076 adults aged 18+ | Cardiovascular | PCORI |
| Blood Pressure (BP) Control Laboratory | Retrospective Observational Study & Intervention Trials | Observational study: 1,737,995 adults aged 18 to 85
Intervention trials: 24 clinics; 2,101 adults aged 18+ |
Cardiovascular | PCORI |
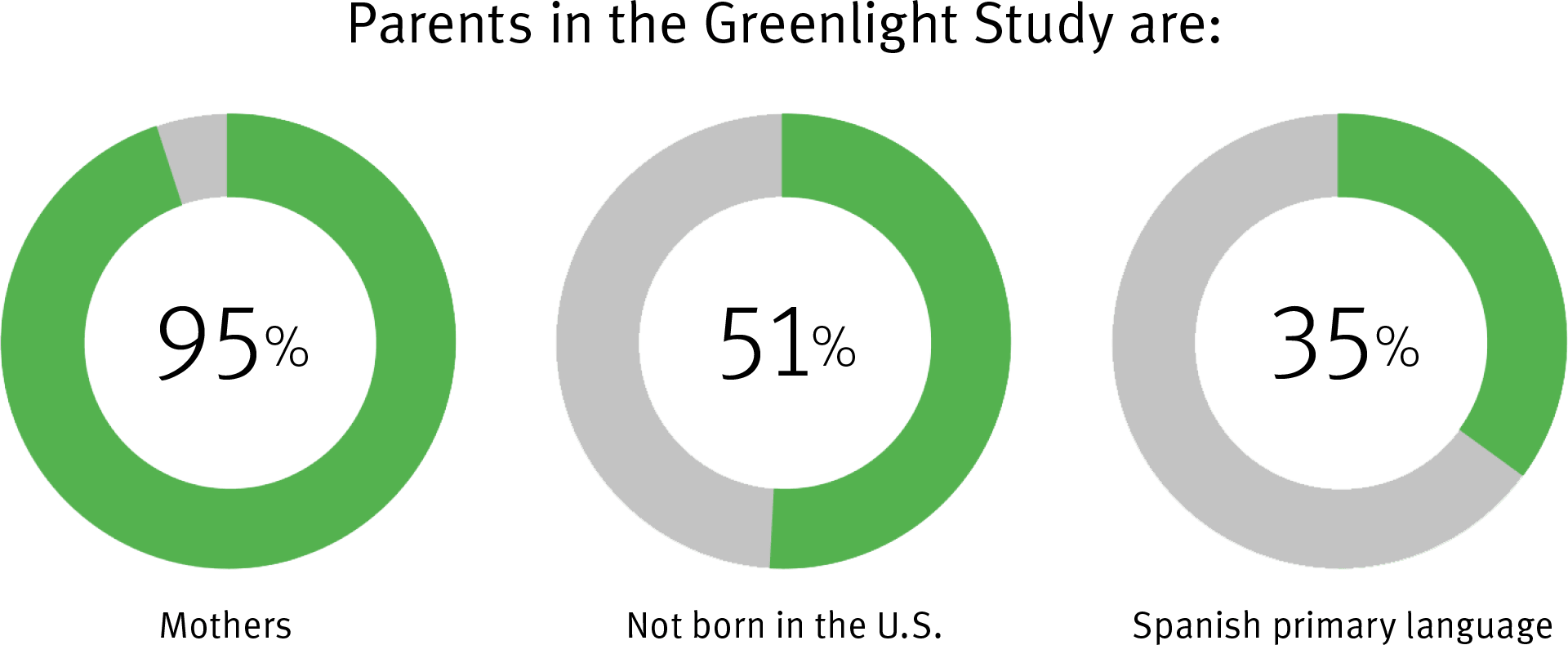
| Comparing Two Ways to Promote Healthy Weight Gain and Prevent Obesity in Early Childhood (Greenlight Plus) | Intervention Trial | 900 English- and Spanish-speaking parent-infant pairs
|
Metabolic Disorders | PCORI |
| Medications and Weight Gain in PCORnet: The MedWeight Study | Prospective Observational Study | Separate analyses include 52,309 to 1,074,314 adults aged 20 to 84 and 74,005 children aged 10 to 19 years | Metabolic Disorders | NIH |
| Neuroendocrine Tumors – Patient Reported Outcomes (NET-PRO) |
Prospective Observational Study | 2,539 adults aged 18+ | Oncology; Rare Diseases | PCORI |
| Pediatric KIDney Stone (PKIDS) Care Improvement Network | Prospective Observational Study | 1,290 youth aged 8 to 21 years
|
Nephrology | PCORI |
| Pragmatic Evaluation of Events and Benefits of Lipid-lowering in Older Adults (PREVENTABLE) | Intervention Trial | Up to 20,000 older adults aged 75+ | Cardiovascular | NIH |
| Provider-Targeted Behavioral Interventions to Prevent Unsafe Opioid Prescribing for Acute Non-Cancer Pain in Primary Care | Intervention Trial | 22,616 adults aged 18+ | Opioid use and pain | PCORI |
Case Study Details
Click on a study name to learn more about its goals, design, results, and how the research teams used the PCORnet infrastructure to successfully conduct their studies.
Get Started with PCORnet
 Looking for more inspiration? Check out the PCORnet® Studies page and Intro to PCORnet to learn more about the wide range of research activities the PCORnet infrastructure can support.
Looking for more inspiration? Check out the PCORnet® Studies page and Intro to PCORnet to learn more about the wide range of research activities the PCORnet infrastructure can support.
All the research teams included in this section started with the same step: reaching out. When you are ready to get started, contact the PCORnet® Front Door. Our team of experts will be happy to guide you in accessing PCORnet resources to achieve your study goals.
While it is best to contact the PCORnet® Front Door early in your study planning to make the most of PCORnet resources, such as assistance with community engagement, the PCORnet® Front Door team works with studies at any stage.









 The
The 
